 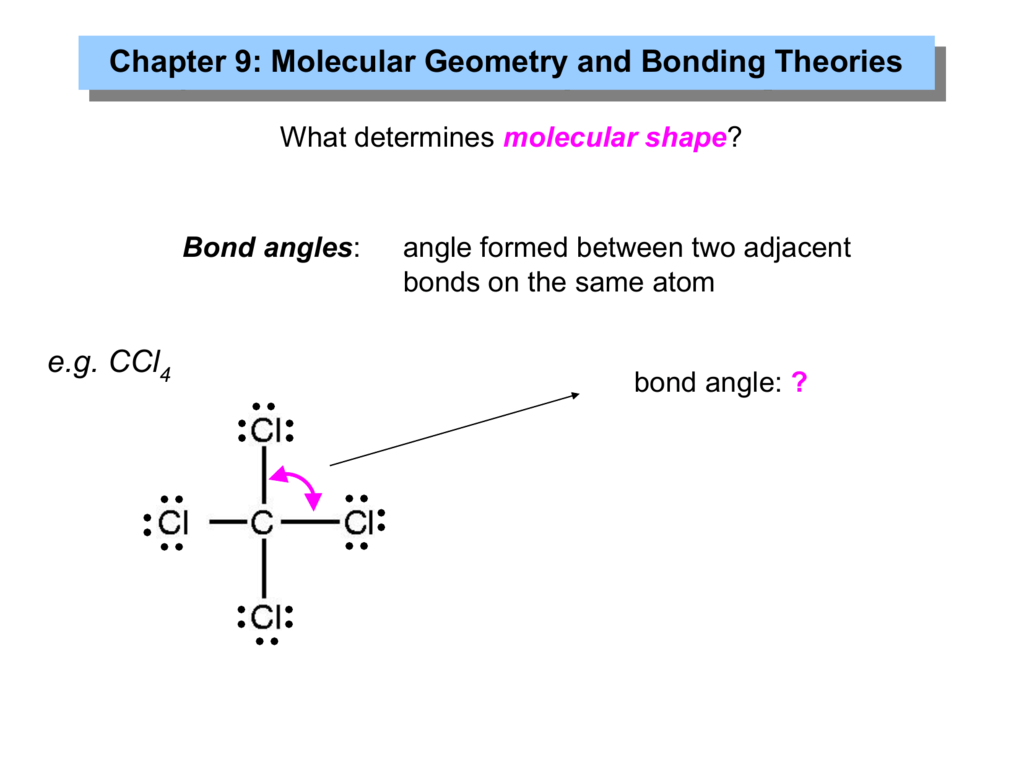
Due to this symmetry, tetrachloromethane is said to be non-polar, even though it contains polar bonds. In case of CCl4, the charges are distributed uniformly around the central carbon atom. Molecules having their charges unevenly distributed are said to be polar in nature, whereas, those molecules whose charges are evenly distributed are non-polar. When it comes to polarity of a compound, we must understand the fact that it is directly related to its structure and shape. The atom of each element present in this compound is stable, in turn, making the compound quite stable. Considering the four atoms of chlorine, each atom will have 6 electrons around it. k2chemistryclass ctc carbontetrachloride shape geometry bondangle bonding hybridization chemistryformula chemistry compound lewisdotstructure ch. The remaining 24 atoms must be distributed in the structure in such a way that all the atoms have 8 electrons in their valence orbital. Since there are no lone pairs on the central carbon atom, the electron group geometry will be determined by the arrangement of the four bonding electron groups. The central carbon atom forms four single bonds with the chlorine atoms. Total valence electrons available for C2H4 lewis structure 42 + 14 12 valence electrons C2H4 has two carbon and 4 hydrogen atom 2. CCl4 has a central carbon atom surrounded by four chlorine atoms. The number of electrons around the carbon atom is 8. And hydrogen only needs one electron to complete its valence shell. In CCl4 (carbon tetrachloride), the central atom is carb. Carbon dioxide (CO 2) is another linear molecule, consisting of two O-C bonds that are 180 degrees apart. An example of a molecule with this geometry is CH 2 CCH 2, which has two H 2 C-C bonds forming a 180-degree angle. Total number of electrons in the compound = Total number of valence electrons of (carbon + chlorine) Total number of electrons in the compound = (1 X 4) + (4 X 7) = (4 + 28) = 32 Question: Part B Select the correct hybridization for the central atom based on the electron geometry CCl4 S1 O sp Submit Request Answer Part C. AX 2 - The two-electron domain structure produces a linear molecule with electron groups 180 degrees apart. The carbon atom shares 1 electron each to form one covalent bond with every chlorine atom to achieve its octet. This implies that carbon requires 4 electrons and chlorine requires 1 electron to satisfy its octet. Thus, carbon and chlorine have 4 and 7 valence electrons, respectively. As we already know, the atomic number of carbon atom is 6, and that of chlorine is 17. 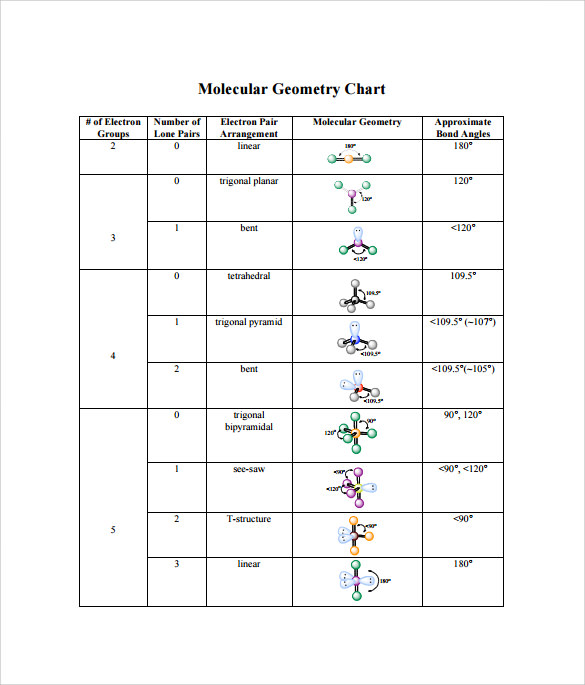
Carbon forms one covalent bond with each chlorine atom, which is electronegative in nature. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
